Introduction
Chemical ionisation mass spectrometry (CI-MS) is a powerful analytical technique used to identify and quantify the composition of chemical compounds. In CI-MS, ion-molecule reactions play a crucial role in the formation of ions that are subsequently detected and analyzed. This article aims to provide an overview of ion-molecule reactions in chemical ionisation mass spectrometry, their significance, and their impact on the analysis of compounds.
Understanding Ion-Molecule Reactions
In chemical ionisation mass spectrometry, ion-molecule reactions occur when a reagent gas is introduced into the ion source of the mass spectrometer. The reagent gas molecules interact with the sample molecules, leading to the formation of ions. These ions are then separated and detected based on their mass-to-charge ratio.
Significance of Ion-Molecule Reactions
Ion-molecule reactions play a crucial role in CI-MS for several reasons. Firstly, they enhance the sensitivity of the technique by increasing the ionization efficiency. The reagent gas molecules act as a source of ions, allowing for the detection of compounds that may have low ionization potentials or are difficult to ionize using other ionization methods.
Secondly, ion-molecule reactions can provide structural information about the sample molecules. The reactions can lead to the formation of specific product ions that can be used to identify the functional groups or the structure of the compound. By analyzing the mass spectra obtained from ion-molecule reactions, researchers can gain insights into the composition and structure of the analyzed compounds.
Types of Ion-Molecule Reactions
There are several types of ion-molecule reactions that can occur in CI-MS. One common type is proton transfer reactions, where a proton is transferred from the reagent gas molecule to the sample molecule. This results in the formation of a protonated molecular ion, which can be detected and analyzed.
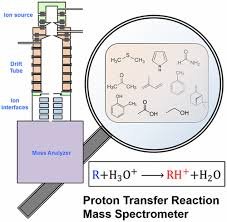
Another type of ion-molecule reaction is charge transfer reactions, where an electron is transferred from the reagent gas molecule to the sample molecule. This leads to the formation of a radical cation, which can also be detected and analyzed.
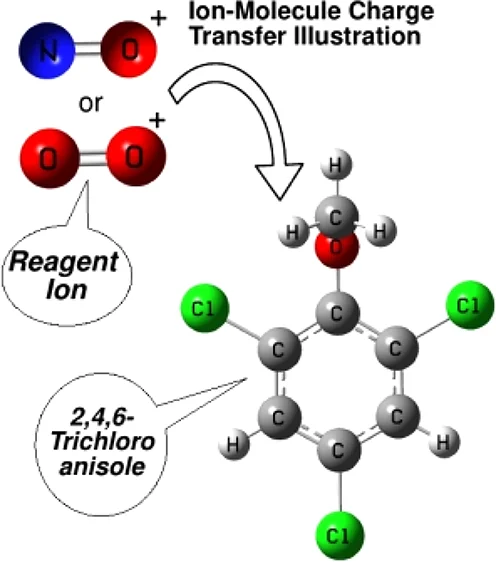
In addition to proton transfer and charge transfer reactions, other ion-molecule reactions such as hydride transfer and adduct formation can also occur in CI-MS. These reactions further expand the range of compounds that can be analyzed using the technique.
Factors Affecting Ion-Molecule Reactions
Several factors can influence the occurrence and efficiency of ion-molecule reactions in CI-MS. One important factor is the choice of reagent gas. Different reagent gases have different ionization potentials and affinities for proton or electron transfer, leading to variations in the ionization efficiency and the types of ions formed.
The pressure and temperature of the ion source can also affect ion-molecule reactions. Higher pressures and temperatures can promote more collisions between the reagent gas and the sample molecules, increasing the likelihood of ion formation.
The nature of the sample molecules, such as their ionization potentials and reactivity, can also impact ion-molecule reactions. Compounds with higher ionization potentials may require more energetic collisions to undergo ionization, while highly reactive compounds may undergo additional reactions, leading to the formation of multiple ions.
Conclusion
Ion-molecule reactions are fundamental to chemical ionisation mass spectrometry and play a crucial role in the analysis of compounds. They enhance the sensitivity of the technique, provide structural information about the sample molecules, and expand the range of compounds that can be analyzed. Understanding the factors that influence ion-molecule reactions allows researchers to optimize the CI-MS method for specific applications, leading to more accurate and reliable results.
Leave a Reply